
USE CASE
Clinical study design and clinical trials are all about spotting the right opportunities at the right time. Depending on your role in the process, this can take various forms, from figuring out which treatments are worth setting up trials for to determining which sites to partner with, and everything in between. With its unique semantic search capabilities and data science intelligence, DISQOVER opens up a whole new range of possibilities.
With DISQOVER, researchers and study designers gain access to a wealth of data from disparate sources that can be easily compared and visualized insightfully. This revolutionizes several main activities, such as:
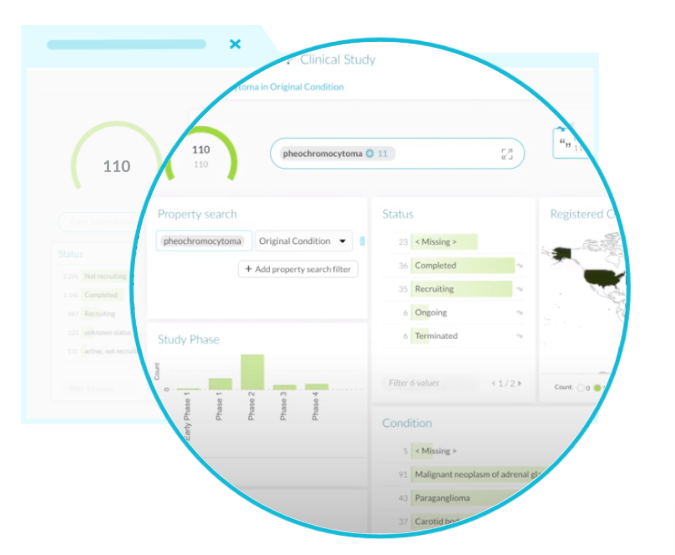
Integrates data from a range of sources and automatically recognizes studies reported multiple times and merges the information
Standardizes filter criteria across databases – e.g., trial phase – and maps these to concepts for further contextual enrichment – including intervention type, sponsor, study site and characteristics, etc.
Allows users to 'backtrack' links to identified concepts, including indication, intervention, study site, sponsor, and investigator.
Creates comprehensive dashboards that offer insights from a chosen point of view, e.g., the disease, the site, the company, or the treatment.
DISQOVER is built upon intelligent semantic search and filter technologies to make this possible. These enable users to reduce a large set of clinical studies and other data points into a manageable subset. Under the hood is a powerful engine that…
Demo video
In this demo, you will discover how to find clinical study sites related to a specific subset of clinical studies. The integration and harmonization of multiple clinical study repositories make finding insights into the 500.000+ available clinical studies fast and easy.
Experience a live walkthrough of DISQOVER with one of our experts, and get to see how our powerful knowledge discovery platform can help you accelerate your drug development activities.
© 2026 ONTOFORCE All right reserved