Have an impact on every part of the value chain

WEBINAR ON DEMAND
The pharmaceutical industry faces many data-related challenges. From data fragmented in silos, to metadata integration and standards management, these are just a few of the issues facing an industry in which timelines are tight, competition is fierce, and patient safety is of paramount importance.
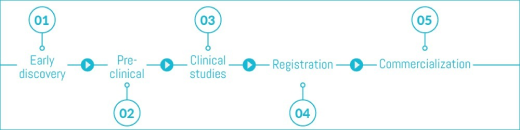
The clinical research phases are critical milestones in drug development. Being able to stay attuned to what’s happening in clinical trials is important in decision-making. Additionally, information about the clinical competitive landscape is spread over different clinical trial registry databases managed by regulatory authorities. Getting a complete competitive overview requires combining and harmonizing this data. In this webinar, learn how DISQOVER enables you to do just that.
Search, filter, compare and make the right action based on data.
.png)
© 2024 ONTOFORCE All right reserved